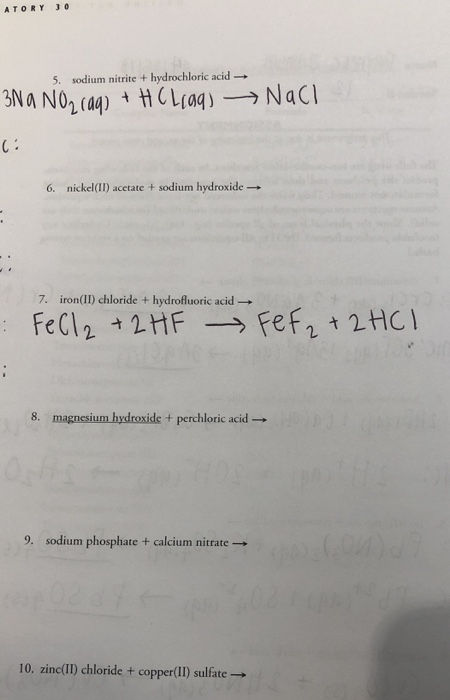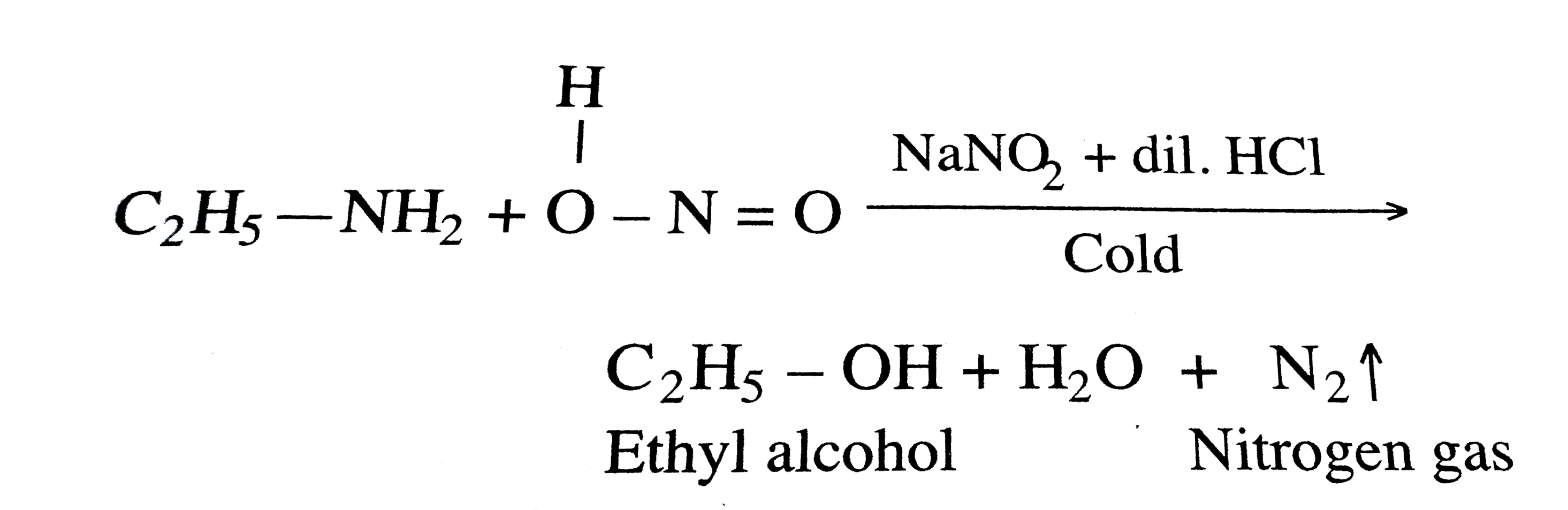
What will be the action of the mixture of sodium nitrite and dilute hydrochloric acid on : (a ) Ethyl amine (b ) Aniline (c ) Triethyl amine
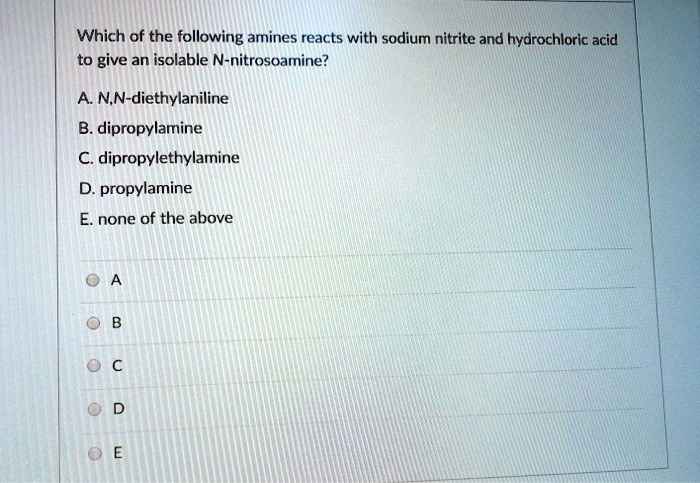
SOLVED: Which of the following amines reacts with sodium nitrite and hydrochloric acid to give an isolable N-nitrosoamine? A NN-diethylaniline B. dipropylamine dipropylethylamine D. propylamine none of the above

Reagents and reaction conditions. a Sodium nitrite, hydrochloric acid.... | Download Scientific Diagram

What will be the action of the mixture of sodium nitrite and dilute hydrochloric acid on : (a ) Ethyl amine (b ) Aniline (c ) Triethyl amine

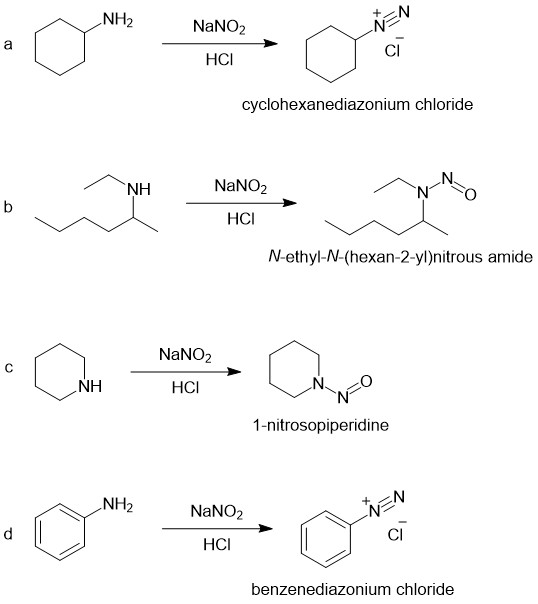
What is the action of sodium nitrite and hydrochloric acid on ethanamine, N - ethylethanamine and N, N - diethylethanamine?

OneClass: Write an equation between sodium nitrite and hydrochloric acid and name the products. Are t...

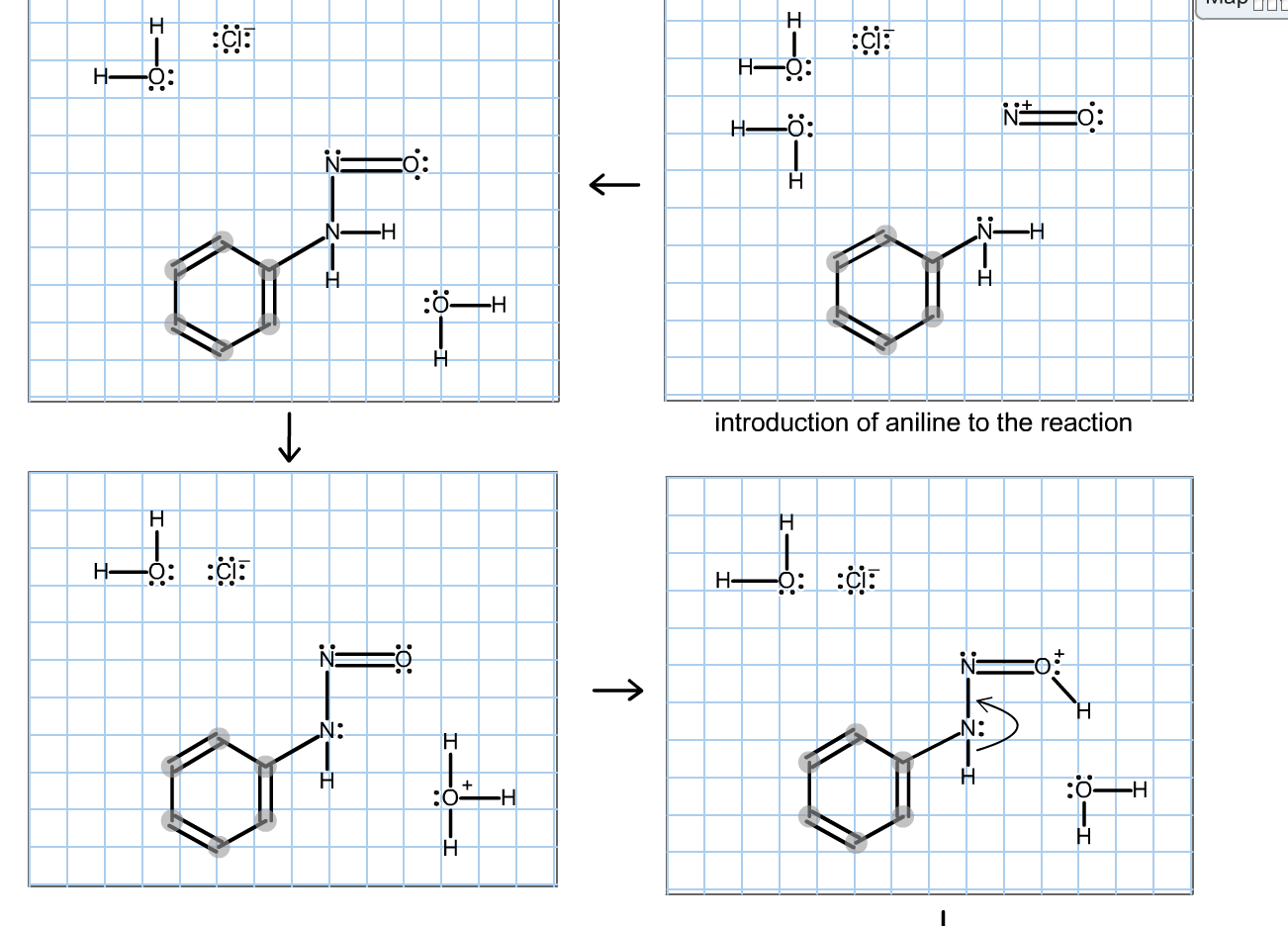
In the diazotisation of aniline with sodium nitrite and hydrochloric acid, an excess of hydrochloric acid is used primarily to :

1) Write an equation between sodium nitrite and hydrochloric acid and name the products. Are the products stable? 2) Write an equation for the formation of p-Iodonitrobenzene starting from p-nitroanil | Homework.Study.com

In the diazotasation of aniline with sodium nitrite and hydrochloric acid, an excess of hydrochloric - YouTube


![Which gas will be evolved when [ CH3CH2NH2 + (CH3)2CHNH2 ] is treated with sodium nitrite and HCl ? Which gas will be evolved when [ CH3CH2NH2 + (CH3)2CHNH2 ] is treated with sodium nitrite and HCl ?](https://dwes9vv9u0550.cloudfront.net/images/10005586/914b2210-e843-4804-ab15-3c7dbfc2348c.jpg)

![Which gas will be evolved when [ CH3CH2NH2 + (CH3)2CHNH2 ] is treated with sodium nitrite and HCl ? Which gas will be evolved when [ CH3CH2NH2 + (CH3)2CHNH2 ] is treated with sodium nitrite and HCl ?](https://dwes9vv9u0550.cloudfront.net/images/4383591/ddb4350c-f6f7-497f-b0a0-48c8b479f1e4.jpg)